|
Singapore-MIT Alliance for Research & Technology |
BioSystems and Micromechanics (BioSyM) Inter-Disciplinary Research Group |
||||||||||||||
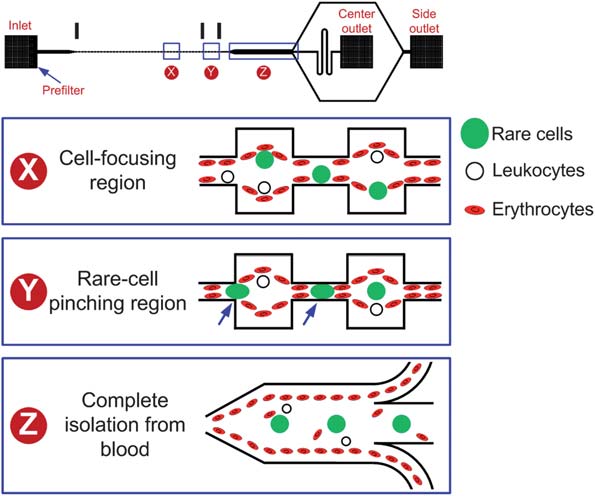
BioSyM's recent publications in "Lab on A Chip" journalSMART BioSyM researchers have recently published some of their work in the prominent journal "Lab on A Chip". The articles address 3 applications of microfluidics. The research work described are results of collaborative efforts between researchers of SMART BioSyM, NUS and MIT. In one titled "Pinched flow coupled shear-modulated inertial microfluidics for high-throughput rare blood cell separation" (Ali Asgar S. Bhagat, Han Wei Hou, Leon D. Li, Chwee Teck Lim and Jongyoon Han, Lab on A Chip, Published on 19 April 2011 on http://pubs.rsc.org | doi:10.1039), the authors describe the application of shear-modulated inertial microfluidics to isolate Circulating Tumor Cells (CTC) from blood. Blood is a complex suspension of cells (40–45% of blood volume) in plasma which plays several key roles including transport of oxygen and nutrients to cells, removal of cellular waste products and providing immunological protection. Red blood cells (RBCs) account for >99% of all hematologic cellular components (5 billion RBCs per millilitre of whole blood) with the remaining <1% consisting of peripheral blood leukocytes (PBL) and platelets. In addition to RBCs and leukocytes, other low abundance cells such as fetal cells, circulating tumor cells (CTCs), stem cells or leukemic cells are also found in peripheral blood of patients, which can potentially be used for various biomedical applications such as disease detection, diagnosis, prognosis, therapeutic treatment monitoring and conducting fundamental scientific studies. However, as the low abundance cells are so rare, it is often necessary to have an enrichment or separation step to efficiently isolate them from blood prior to analysis. This paper describes the application of shear-modulated inertial microfluidics to isolate Circulating Tumor Cells (CTC) from blood.
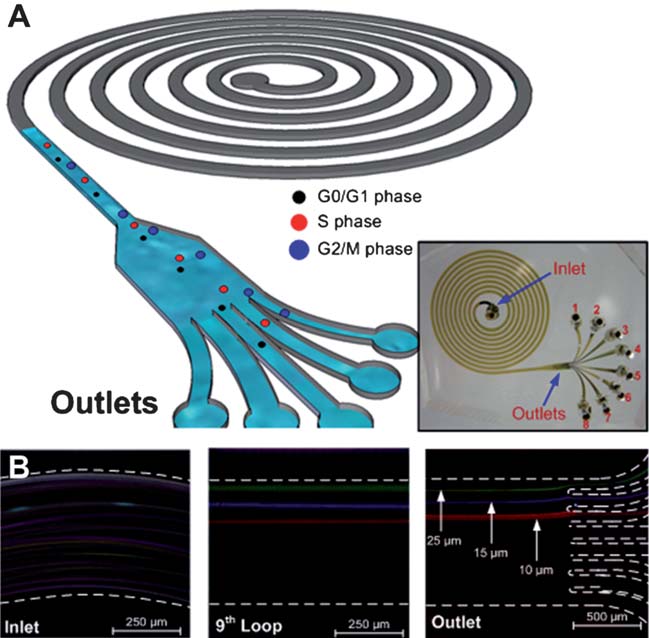
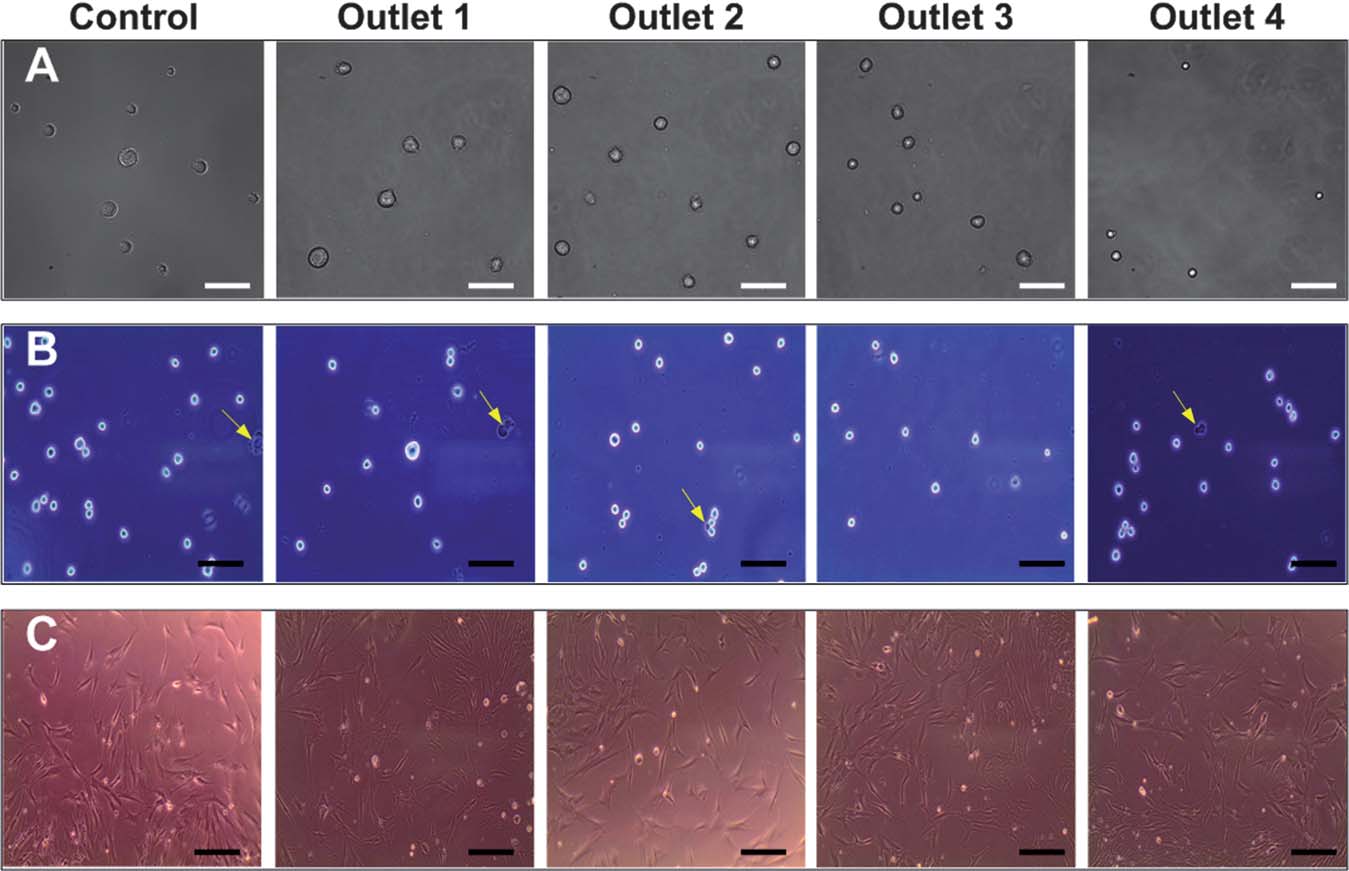
Another paper on "High-throughput cell cycle synchronization using inertial forces in spiral microchannels" (Wong Cheng Lee, Ali Asgar S. Bhagat, Sha Huang, Krystyn J. Van Vliet, Jongyoon Han and Chwee Teck Lim, Lab on A Chip, 11, 1359 (2011)), describes a microfluidics based approach to synchronize cells using inertial forces in spiral microchannels. Cell cycle synchronization is essential for studying cellular properties, biological processes and elucidating genetic regulatory mechanisms and events involved in each phase prior to cell division. In eukaryotic cells, the distinct events leading to proper cell division can be separated into four sequential phases: G1(gap), S (DNA synthesis), G2 (gap) and M (mitosis). As a cell progresses through the cell cycle, it duplicates its hromosomes during the S phase and segregates the chromosomes in the M phase. The use of a highly synchronized population of cells has facilitated the development of a variety of biological systems. For example, via such synchronization of cancer cells, several key oncogenes have been identified and implicated in specific cell cycle checkpoints. Development of cancer therapeutics has thus employed tumor cell synchronization, because anticancer drugs are known to target cells in different phases of the cell cycle. In stem cell therapies that involve nuclear transfer to the host cells, cell cycle synchronization is critical because stem cells in the G0/G1 phase impart higher nuclear transfer efficiency. Thus, there is great interest in developing efficient techniques to rapidly synchronize and to isolate cells at various phases of the cell cycle. This paper describes a microfluidics based approach to synchronize cells using inertial forces in spiral microchannels.
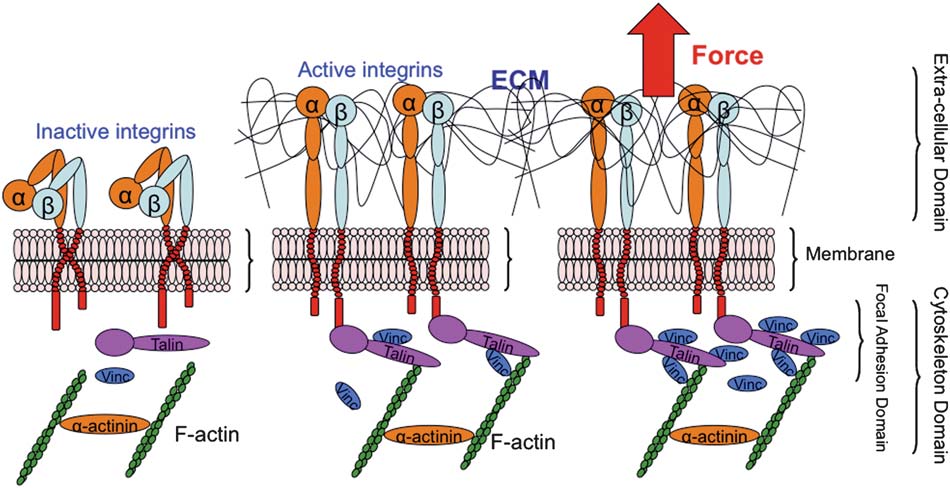
In the 3rd paper, "A microfluidic system with optical laser tweezers to study mechanotransduction and focal adhesion recruitment" (Peyman Honarmandi, Hyungsuk Lee, Matthew J. Lang and Roger D. Kamm, Lab on A Chip, 11, 684 (2011)), a microfluidic device and optical laser tweezers are used to apply mechanical stimuli to cells. In the last decade, several experiments have investigated the importance of mechanosensing and the assembly of focal
complexes at adhesion sites under the application of mechanical force with various ECM proteins. These studies demonstrated that mechanical forces are required for the initiation and Using microfluidics provides convenient means of introducing and positioning cells under controlled and physiological conditions. In this particular instance, a microfluidic device was designed to confine the cells and to expose a portion of the cell membrane to a bead functionalized with an integrin-binding protein, e.g. fibronectin, to emulate ECM-integrin-cytoskeleton linkages, as illustrated below. An optical laser trap is used to facilitate and apply force in a defined direction on the exposed portion of the cell membrane via its receptors. Then, utilizing immunostaining for proteins of interest, i.e. vinculin in our case, the recruitment of protein is studied upon application of an external tensile load. In addition, the mechanical properties of the FA complex can be inferred from the measurement. The main advantages of the proposed method are controllability, physiological environment, introducing beads, ease of numerical simulations, local excitation with defined geometry, and most importantly the capability to directly image protein recruitment, which other techniques such as atomic force microscopy or micropipette aspiration may not offer.
|
|||||||||||||||